Fernando Botto –
ESC 2019 hot line sessions delivered us enough evidence to discuss methods and patient-centered decisions. For instance, dual antiplatelet treatment came up with a new trial in Stable CAD patients, besides the previous CHARISMA with clopidogrel and PEGASUS with Ticagrelor. DAPT trial is not discussed because a different “Stable CAD” scenario, just after 1 year post PCI with dual antiplatelet treatment without complications.
THEMIS trial summary
In patients of 50 years or more with Stable CAD (prior PCI or CABG or coronary estenosis >50% [MI or stroke excluded]) and Diabetes, adding Ticagrelor 90 mg twice daily or Placebo to aspirin, will reduce a composite outcome of CV death, MI or Stroke at 40 months?
https://www.nejm.org/doi/full/10.1056/NEJMoa1908077
Design: international 1:1 RCT, triple-blind (patients, investigators and event adjudicators). Sample size estimation 19.000 assuming a 16% risk reduction with Ticagrelor (power 90%, alpha 5%).
Results: the primary efficacy outcome was 7.7% with Ticagrelor and 8.5% with placebo (HR 0.90, CI 0.81-0.99; p = 0.04). The observed benefit was driven by a lower incidence of MI and stroke without affecting CV death. Contrarily, the primary safety outcome showed 2.2% vs 1% of major bleeding (HR 2.32, CI 1.82-2.94; p<0.001). Regarding intracranial hemorrhage, ticagrelor had also a significant higher incidence (0.7% vs. 0.5%; HR 1.71, CI 1.18-2.48; p= 0.005). NNT (Number Needed to Treat) was 138 and NNH (NN to Harm) 93. The authors concluded that the dual treatment reduced ischemic events at the expense of higher chance of major bleeding, and further, there was a non-significant reduction in an exploratory outcome of “irreversible harm” that included total death, MI, stroke, fatal and intracranial bleeding.
Methodological topics
The target population
Does it make sense a different antithrombotic treatment for a diabetic patient with Stable CAD because she/he had or not a prior MI?
My first concern is about the rationale of the trial… PEGASUS trial included patients with Stable CAD with a prior MI and showed no effect interaction between diabetics and non-diabetics. Thus, scientific treatment inferences should include both subgroups. As PEGASUS, THEMIS trial was focused also on demonstrating any benefit of dual antiplatelet treatment in Stable CAD patients with elevated risk, now including Diabetes without a prior MI. Intuitively, I would have expected a lower risk population in THEMIS than in PEGASUS and why not similar results? (easy to write after knowing the results). In fact, the primary outcome of control group was lower in THEMIS (8.5%) than PEGASUS (9.04%). And thus, similar results!
Confidence intervals, imprecision and clinical decision threshold
Precision of treatment effect relies mostly in sample size and number of primary events that determine the Confidence Intervals (CIs), which represent the range within the truth may lie. Therefore, CIs inform about random error. In THEMIS we have nothing to discuss here because of a large sample size (n=19,220) and enough number of primary MACE (n=1,554) determining narrow CIs. The primary efficacy outcome favoured Ticagrelor with a HR 0.90, CI 0.81-0.99; p = 0.04. However, statistically significant result does not mean clinically significant result for patients.
When you calculate sample size, you may consider a risk reduction with the new treatment, let us say 16% (expected HR of 0.84) like in THEMIS. But, depending of the outcomes importance, frequency and life-threatening of underlying disease, and the complexity, cost and potential SAEs of the new treatment, we should require a minimally important difference (MID) before supporting superiority of the new treatment as a general recommendation.
The observed right margin of CI was 0.99, not crossing the no-effect vertical line and meaning only a 1% of relative risk reduction, statistically significant but almost nothing. Taking into account the higher risk of major bleeding and intracranial hemorrhage observed with ticagrelor, I would have required probably a 10% MID which, far from being the right boundary, it was actually the average result (HR 0,90) favoring efficacy of ticagrelor. We may recall, for instance, IMPROVE IT trial, where adding ezetimibe or placebo to simvastatin after ACS showed a RR of 0.93 (0.89 to 0.99, p=0.01) in the primary efficacy outcome. Since ezetimibe did not show any safety issues compared to placebo, it treats a common disease and cost is low, we may support a minimal clinical benefit in this setting.
Dose issue, modified ITT and lack of adherence
After PEGASUS results determined better risk/benefit profile of ticagrelor 60 mg x2, THEMIS investigators changed the dosis of 90 mg x2 by the 60 mg x2, which finally totalized 76% of the total exposure of ticagrelor. Different sensitivity analyses were performed according to received dose and the number of censoring days after the last dose, demonstrating similar benefit in different scenarios but not all statistically significant. This highlights the fragility of a marginal benefit, as it was discussed before.
To avoid risk of bias and the beauty of randomization, superiority trials should undergo ITT analysis. THEMIS reported a “modified ITT” analysis, which is usually applied to exclude some randomized patients in a justified manner. In THEMIS, 51 patients were excluded before unblinding because the sponsor closed one site due to GCP inconvenience. There are no clear statements to deal with the modified ITT, therefore it might be considered a risk of bias because allows for subjective manipulation of data.
Finally, as in other trials dyspnea and bleeding were the reason of a more frequent permanent discontinuation of ticagrelor in 34.5% of patients compared with placebo in 25.4%. The impact of this difference was also evaluated in sensitivity analyses with no significant influence.
Research in context
(In) Consistency of results
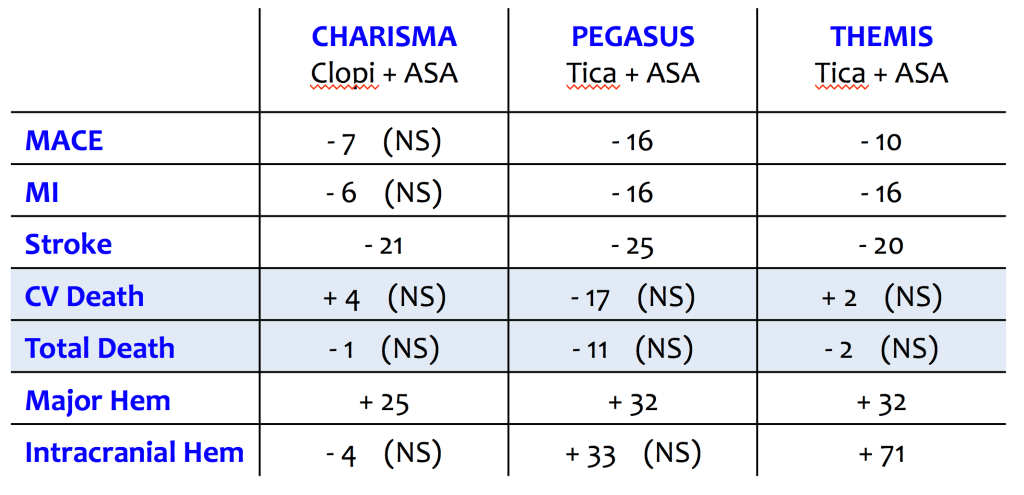
CHARISMA, PEGASUS AND THEMIS demonstrated a consistent benefit in terms of adverse ischemic events (non-significant with clopidogrel and significant with ticagrelor – see Table with relative risks) counterbalanced by a significant higher risk of major bleeding in each trial. Probably, it was no benefit with clopidogrel due to a lower risk population included in CHARISMA but every individual outcome were in the same direction. The consistency of results across the 3 trials reinforces their external validity. If we evaluate only ticagrelor trials, PEGASUS and THEMIS showed practically the same results on primary efficacy and safety outcomes. Importantly, no significant CV and total death differences were found.
Take-home message
Across existing trials in Stable CAD with prior MI or Diabetes adding Ticagrelor to ASA has demonstrated a consistent but minimal benefit in terms of the occurrence of ischemic events (MI or stroke) without impacting CV death. This effect is not clinically important and is counterbalanced by higher major bleedings. Additionally, 1 out of 3 patients have discontinued ticagrelor due to side effects.
It seems to be not “practice changing” in terms of general recommendations. However, it might allow for a “personalization” of treatment in cases with very high ischemic risk or repeated ischemic events and with an estimated low bleeding risk, always discussing preferences and expectations with the patient. Finally, in these cases I would incorporate to the discussion the use of new oral anticoagulants such as low-dose rivaroxaban (2.5 mg x2) plus ASA based on COMPASS trial results, which also increases major bleeding risks but shows a higher impact on ischemic outcomes reduction including CV death. Great topic for a future review.
